Phenols, Alcohols and Carboxylic Acids - pKa Valuesįor oxygen containing organic compounds this is given: pKa (the negative logarithm of the acid dissociation constant), molecular structures, molar weights, density and melting and boiling points. Introduction to pH - the acidic and basic (alkaline) definition. Metals - Corrosion Resistance to Aggresive FluidsĬommon metals and their corrosion resistance to aggressive fluids like acids, bases and more. Specific heats for some common liquids and fluids - acetone, oil, paraffin, water and many more. Values for the negative logarithm of the acid dissociation constant, pKa, of inorganic acids and bases, as well as hydrated metal ions.ĭensities of common liquids like acetone, beer, oil, water and more. 
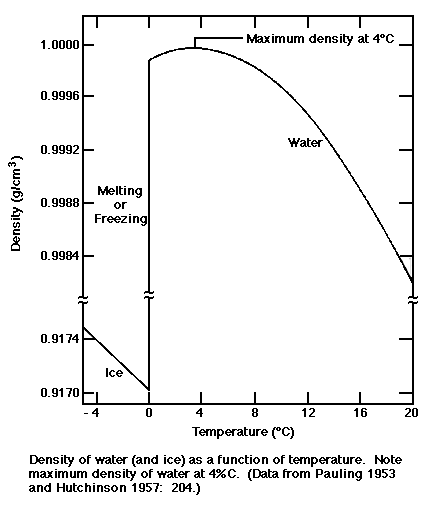
Density of inorganic substances in water is plotted as function of wt%, mol/kg water and mol/l solution. 
Density of acetic acid, citric acid, formic acid, D-lactic acid, oxalic acid and trichloroacetic acid in water is plotted as function of wt%, mol/kg water and mol/l solution.ĭensity of Aqueous Solutions of some Inorganic SubstancesĬhanges in density of aqueous solutions with changes in concentration at 20☌. What is the m of a 5.76 (v/v) solution of ethanol (Note that the density of ethanol is 0.789 g/mL.) The density of an aqueous solution containing 10.0 ethanol by mass is 0.984 g/mL. Molweight, melting and boiling point, density, pKa-values, as well as number of carbon and hydrogen atoms in molecules are given for 150 different alcohols and acids.ĭensities of Aqueous Solutions of Organic AcidsĬhanges in density of aqueous solutions with changes in concentration at 20☌. The density of ethanol is 0.78 g/mL, the density of water is 1.0 g/mL, and the density of the solution is 0.968 g/mL. PH values of acids like sulfuric, acetic and more.Īlcohols and Carboxylic Acids - Physical Data color change for acid and base indicators - together with pKa and structures of the indicators. Read more about how to correctly acknowledge RSC content.Material properties of gases, fluids and solids - densities, specific heats, viscosities and more. Please go to the Copyright Clearance Center request page. In a third-party publication (excluding your thesis/dissertation for which permission is not required) 
If you want to reproduce the whole article If you are the author of this article, you do not need to request permission to reproduce figuresĪnd diagrams provided correct acknowledgement is given. ( a ) What is the coefficient of volume expansion of the container ( b ) What is the most likely material of the container Density of water at 60☌ is 0.98324 g/mL. When the container and the water are heated to 60☌, 0.35 g of water is lost. Temperature Density of Ethyl Alcohol aqueous solutions. Density of inorganic substances in water is plotted as function of wt, mol/kg water and mol/l solution. Provided correct acknowledgement is given. (II) It is observed that 55.50 mL of water at 20☌ completely fills a container to the brim. Changes in density of aqueous solutions with changes in concentration at 20C. If you are an author contributing to an RSC publication, you do not need to request permission To request permission to reproduce material from this article, please go to the The results demonstrate the potential of dual-modification design using solution-based processes to enable sustainable energy technologies.ĭual modification on hematite to minimize small polaron effects and charge recombination for sustainable solar water splitting The engineered photoanode increased photocurrent from 0.7 mA cm-2 for pristine hematite up to 4.5 mA cm-2 at 1.23V and beyond 6.0 mA cm-2 when applying an overpotential of 300 mV under simulated sunlight illumination (100 mW cm-2). The solution-based method simultaneously induces Al3+ doping of hematite crystal lattice while Zr4+ forms interfacial excess, creating a single-phased homogenous nanostructured thin film. Here we develop a synthetic strategy to leverage earth-abundant Al3+ and Zr4+ in a dual-chemical modification to synergistically minimize small polaron effects and interfacial charge recombination. However, major challenges exist in improving charge density and minimizing charge recombination rates for a competitive photoelectrochemical performance based on hematite without compromising sustainability aspects. Hematite nanostructures are strong candidates for the development of sustainable water splitting technologies.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
